Identification of Unknowns
As a commercial testing laboratory, we get requests daily from individuals and businesses wanting to know “what is this?” Unfortunately we still don’t have that magic box to put the material in that will spit out the unequivocal complete answer, however armed with a bit of analytical chemistry and a lot of specific magic boxes, we can often times paint the complete picture of ”what is this?”
The first question we tend to ask in the lab is how much of this do you need to know? The answer to that question will govern the choice of screening techniques utilized. How simple is the material? Is this simply a single component reaction product? Or to we have a mixture or solution of several components? Are there biologics involved? Some biologics might present as simple organic molecules with mineral components, however the methods of characterization are completely unique in analytical chemistry.
As a service provider we get varying levels of sophistication in the inquiries, and it is our job to assess the target objectives with the information provided. The inquiries will range from the person who thinks there is a magic box and can get the answer with $100, to the chemist who is trying to fully deformulate a complex mixture or consumer product. In addition, the amount of information coming with the material can be the number one influencer of techniques to utilize in characterization. With all of the variables presented, we will simplify this discussion to one scenario as follows: That of a residue identification with no information as to the process chemistry.
Case: Unknown Residue Identification
The client has isolated a visible residue from a surface and transferred to a plastic bag. The residue was on an outside surface that was painted steel. The client did an adequate job or isolating the residue from the surface with a rigid instrument but has absolutely no history or knowledge of the chemistry going on in the vicinity. The client does not want to spend a lot of money on this identification but has a curiosity factor. Is this material dangerous/hazardous? Is this residue going to be detrimental to the substrate it is on? Did it come from the substrate? How did it get here?
With those questions in mind we formulate a plan of attack that will general involve a two stage screening approach. We will typically utilize both qualitative elemental and molecular spectroscopy techniques. For the elemental spectroscopy component, we will transfer a portion of the residue to a mount and analyze by Scanning Electron Microscopy (SEM) and Energy Dispersive X-ray Spectroscopy (EDS). For the SEM portion, we get a microscopic view into the bulk matrix to see what the microscopic morphology is. For instance, we might see a lot of fibrous material in an amorphous “gel” with several round particles comprising the bulk. This imaging will be our first indication of the relative homogeneity of the matrix. If we do the characterizations as a “whole”, we might be looking at too much information to properly conclude as to the components. If we start to deal with these morphological artifacts independently, it is much easier to conclude that yes this residue is a heterogeneous mixture of three different and unique components.

In conjunction with the SEM, we can utilize this morphological information to also “target” specific areas for elemental composition, or if need be, do further separations of the individual components to “clean them up” for the elemental analysis.
Whether bulk or targeted elemental composition is warranted, both tasks can be accomplished in the SEM chamber. After the elemental spectra are collected we have an almost clear picture as to the elemental constituents along with relative levels of those constituents. We must realize the limitations of the techniques that we are only capable of looking at elements with atomic number greater than five and present at a level of greater than 0.1% by weight. Therefore if we are suspect significant lithium or beryllium concentrations, or other light cations such as ammonium presence, we must take this into consideration in our data analysis.

After elemental spectroscopy, we will turn our attention over to molecular spectroscopy. In most cases, this will involve Fourier Transform Infrared Spectroscopy (FTIR). We can also apply the same morphological criteria as in the SEM. If we know we are dealing with heterogeneous morphologies we can target those morphologies in a similar fashion using microscopic FTIR. It is also at this point in the analysis, we will get a clear picture as to if and how much water might be present.
As water is a significant interferent in FTIR analysis, the first pass spectrum will let us know about the possible level of water present, which will also be a significant finding in the analysis. After the initial pass with high water presence indicated, we would then dewater the sample to yield better spectroscopic detail regarding the non-water components.

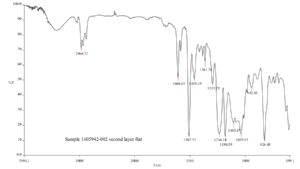
After all spectra and morphological information are obtained, we can then sit down with the data and put it all together. Although elemental and molecular spectroscopy are independent techniques, they will provide both unique and complementary information in the data analysis. Although elemental spectroscopy is not a compound identification technique, we can often utilize the quantitation from the elemental spectroscopy to suggest likely compounds due to mass balance and stoichiometry routines. In addition, quality FTIR spectra can be search against vast maintained standard libraries.
After data analysis, either ambiguous spectral information or need for further speciation and quantitation of one or more component may be deemed necessary. Again, this is the task of the scientist to determine these points and possibly suggest additional techniques to supplement the data provided, such as X-ray Diffraction (XRD) to identify and quantify crystalline inorganic materials present or liquid or gas chromatography to identify and quantify specific organic compounds present. Either way, for unknown residue identification, the most efficacious and cost effective starting point is elemental and molecular spectroscopy.
About the author:
Lloyd Kaufman is the Director of Materials Sciences at RTI Laboratories, and has been performing successful unknown residue identifications for a good chunk of 25 years.
